The horse’s stifle is often considered particularly complex because of the large number of associated intraarticular and periarticular soft tissue structures [1]. Furthermore, injuries to the soft tissues oftentimes occur in conjunction with osseous (boney) damage that leads to joint instability and contributes to the development of osteoarthritis (OA).
Meniscal injuries in the stifle are not uncommon and the majority occur in the medial femorotibial articulation. Horses with meniscal injuries may present as severe acute injuries or as more chronic low grade lameness’s. Radiographs provide little direct evidence of pathologic changes in soft tissues and ultrasound remains the most commonly useful diagnostic imaging modality for veterinarians.
Fibre damage in the meniscus can present as discrete tearing or more diffuse injury, with areas of hypoechoic or heterogenous appearance on ultrasound. Generally the prognosis in sport horses with meniscal injuries has been considered guarded and current treatments in both humans and horses has revolved around arthroscopic debridement combined with intraarticular treatments. However, the benefits of arthroscopic debridement has recently been questioned in human medicine [2], and in horses some authors report a return to athleticism when using intraarticular medications such as stem cells alone [1].
Here we report on a case of medial meniscal injury in a horse treated with intraarticular 2.5% polyacrylamide hydrogel alone and that results in a favourable outcome.
Case History
A 9 year old showjumper, competing in 1.35m classes presented with an acute onset moderate-severe left hindlimb lameness.
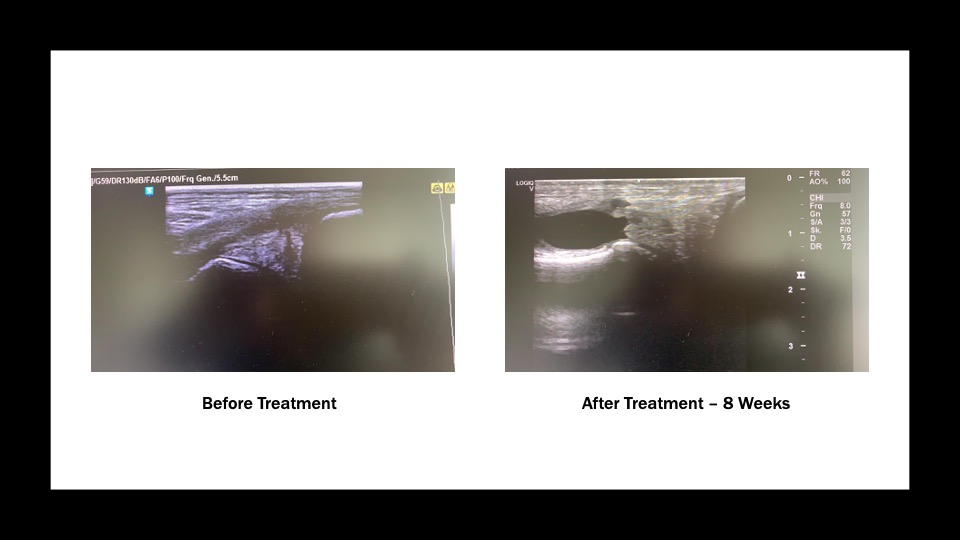
Clinical examination revealed pain on flexion test and effusion of the left medial femorotibial joint (MFTJ). Intraarticular analgesia of the left MFTJ was performed using 20mls of a 2% mepivacaine hydrochloride injected via the medial approach. The lameness was eliminated after 10 minutes. Radiographs (lateral, caudocranial and caudo-45 degrees lateral-craniomedial oblique views) were unremarkable. Ultrasound examination was performed and confirmed a longitudinal tear of the medial meniscus of the left stifle (Figure 1.)
Treatment and outcome
The left MFTJ was medicated with 2mls (50mg) of 2.5% polyacrylamide hydrogel (Arthramid® Vet). The horse was strictly rested for 4 weeks, being restricted to a small yard during the day and boxed at night. The horse then commenced in-hand walking starting at 10 minutes per day and building up by an additional 5 minutes each subsequent week for another 4 weeks (30 minutes hand walking by week 8 after injury).
At 8 weeks the horse was lame free and repeat ultrasound examination showed increased and more homogeneous echogenicity in the area of the initial tear of the medial meniscus (Figure 2). The horse continued its ascending exercise program over a further 8 week period, by the end of which the horse started light jumping exercise. At 6 months post injury, the horse was still lame free and competing at the same level as prior to the injury.
Discussion
Meniscal injuries in horse generally carry a guarded prognosis. The ideal therapeutic agent for stifle lameness in horses would be one that not only alleviates the symptoms of lameness, but also provides a positive effect on the articular soft tissue components at the same time, resulting in a stabilisation or even repair of the pathologic processes in the affected joint.
2.5% Polyacrylamide hydrogel (2.5% PAAG) is an inert, non-pyrogenic and neuro-innocuous polymer gel consisting of 97.5% sterile water and 2.5% cross-linked polyacrylamide hydrogel. Upon intraarticular injection it acts as a bio-scaffold, supporting cell migration and tissue integration to produce a permanent, stable augmentation effect on the soft tissues due to its constant molecular water exchange with the host tissue.
A number of studies have now shown the effective treatment of joint lameness in horses using 2.5% PAAG. Distal interphalangeal (coffin), metacarpophalangeal (front fetlock), metatarsophalangeal (hind fetlock) , carpal (knee) and tarsocrural (hock) joints have all been treated, with the efficacy of a single intra-articular dose of 2.5% PAAG lasting up to 24 months post-administration [3-7]. No case reports have been published to date on the use of 2.5% PAAG in the treatment of stifle lameness in horses due to medial meniscal ligament tears. More extensive and randomised clinical trials are necessary to confirm its relative efficacy and to compare that against arthroscopy alone and/ or stem cell treatments.
Conclusion
2.5% PAAG has in this case offered the veterinary clinician a viable therapeutic alternative for the treatment of stifle lameness in horses due to soft tissue meniscal tears.
Reference
- Barret M F and Frisbie D D. Stifle. In: McIlwraith CW, Frisbie DD, Kawcak CE and van Weeren PR, editors. Joint Disease in the Horse 2nd Edition, Missouri: Elsevier, 2016, p. 354-371
- Siemieniuk R, Harris I, Agoritsas T, Poolman R W, Brignardello-Petersen R, Van de Velde S, Buchbinder R, Englund M, Lytvyn L, Quinlan C, Helsingen L, Knutsen G, Rydland Olsen N, Macdonald H, Hailey L, Wilson H M, Lydiatt A and Kristiansen A. Arthroscopic surgery for degenerative knee arthritis and meniscal tears: a clinical practice guideline. Br J Sports Med2018 52: 313
doi: 10.1136/bjsports-2017-j1982rep - Janssen I, Koene M and Lischer C. Intra-articular application of polyacrylamide hydrogel as a treatment of osteoarthritis in the distal interphalangeal joint: case series with 12 horses. Pferdeilkunde 2012; 28(6): 650-656.
- Tnibar A, Schougaard H, Koene M and Markussen B. A controlled clinical trial on the efficacy of an intra-articular polyacrylamide hydrogel in horses with osteoarthritis. Proceedings of the European College of Veterinary Surgeons Annual Scientific Meeting 2014; 3-5th July, Copenhagen, Denmark.
- Tnibar A, Schougaard H, Camitz L, Rasmussen J, Koene M, Jahn W and Markussen B. An international multi-centre prospective study in the efficacy of an intraarticular polyacrylamide hydrogel in horses with osteoarthritis: a 24 months follow-up. Acta Veterinaria Scandinavia 2015; 57:20.
- Bathe, AP, Read RM and Briggs C Intra-articular polyacrylamide hydrogel for the treatment of 20 horses with non-responsive osteoarthritis of the interphalangeal joints: a prospective study. Veterinary Orthopaedic Society 43rd Annual Conference Abstract 2016; USA.
- de Clifford LT, Lowe JN, McKellar CD, Bolwell C and David F. Use of a 2.5% cross-linked polyacrylamide hydrogel in the management of joint lameness in a population of flat racing Thoroughbreds: A pilot study. Journal of Equine Veterinary Science 2019; 77: 57-62.